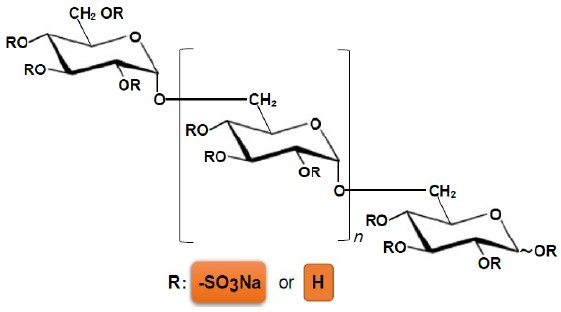
CAS No. : 9011-18-1
CAS No. : 9011-18-1 |
| This page introduces our dextran sulfate (DS) for drug substances, raw material of the medical devices and reagents. |
| Brand name | Molecular weight (Dextran) |
Molecular weight (DS: *) |
Molecular weight (DS: **) |
Purpose |
|---|---|---|---|---|
| Dextran Sulfate Sodium Sulfur 18 | ca. 1,500-1,900 | ca. 5,000 | ca. 4,000 | Drug substance Additives Raw materia of Medical Devices |
| Dextran Sulfate Sodium Sulfur 5 | ca. 1,500-1,900 | ca. --- | ca. --- | Raw material of Medical Devices |
| DS 500 | ca. 500,000-700,000 | ca. --- | ca. --- | Reagent (hybridization) |
| DST-H | ca. 1,500-2,400 | ca. 5,300 | ca. 6,000-7,000 | Diagnostics (cholesterol assay) |
|
(*) Measured by Laser Light Scattering Apparatus
(**) Caluculated from Molecular Weight of Dextran and the Degree of Substitution of Sulfate |
| Test items | The Japanese Pharmacopoeia (18th Ed.) | ||
|---|---|---|---|
| Dextran Sulfate Sodium Sulfur 18 | Dextran Sulfate Sodium Sulfur 5 |
||
| Description | White to light yellowish white powder, odorless, saline taste | White to light yellowish white powder, odorless, saline taste | |
| Freely soluble in water and practically insoluble in ethanol(95) and in ether | Freely soluble in water and practically insoluble in ethanol(95) and in ether | ||
| Hygroscopic | Hygroscopic | ||
| Identification | Toluidine blue | Positive | Positive |
| Anthrone | Positive | Positive | |
| Sodium salt | Positive | Positive | |
| Optical rotation | [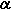 ]D: +90.0 - +110.0° (20°C, 1.5 g/25 mL, 100 mm) ]D: +90.0 - +110.0° (20°C, 1.5 g/25 mL, 100 mm) |
[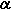 ]D: +135.0 - +155.0 ° ]D: +135.0 - +155.0 ° (20°C, 1.5 g/25 mL, 100 mm) |
|
| pH | 5.5 - 7.5 (1.0 g/20 mL) | 5.0 - 7.5 (1.0 g/20 mL) | |
| Purity | Clarity of solution | --- | Clear (2.5 g/50 mL), Absorbance:Not more than 0.090 (at 420 nm) |
| Chloride | Not more than 0.106 % | Not more than 0.106 % | |
| Sulfate | Not more than 0.480 % | Not more than 0.240 % | |
| Heavy metals | Not more than 20 ppm | Not more than 20 ppm | |
| Arsenic | Not more than 2 ppm | Not more than 2 ppm | |
| Sulfur content | 15.0 % - 20.0 % | 3.0 % - 6.0 % | |
| Loss on drying | Not more than 10.0 % | Not more than 10.0 % | |
| Intrinsic viscosity | 0.020 - 0.032 dL/g | 0.030 - 0.040 dL/g | |
| Test items | Japanese Standards of Quasi-drug Ingredients 2021 | |
|---|---|---|
| Description | White to light yellowish white powder, odorless | |
| Identification | Toluidine blue | Positive |
| Anthrone | Positive | |
| Sodium salt | Positive | |
| IR absorption spectrum | Specific absorption is observed | |
| Optical rotation | [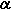 ]D: +85.0 - +115.0° (20°C, 1.5 g/25 mL, 100 mm) ]D: +85.0 - +115.0° (20°C, 1.5 g/25 mL, 100 mm) |
|
| pH | 5.5 - 7.5 (1.0 g/20 mL) | |
| Purity | Chloride | Not more than 0.36 % |
| Sulfate | Not more than 0.48 % | |
| Heavy metals | Not more than 20 ppm | |
| Arsenic | Not more than 2 ppm | |
| Clarity of solution | Colorless to light yellow, clear | |
| Sulfur content | 15.0% - 20.0% | |
| Loss on drying | Not more than 10.0 % | |